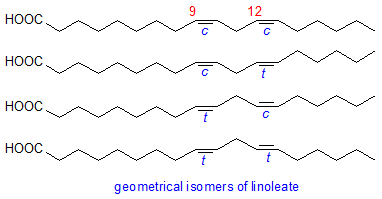
Cis 9 trans 11 isomers

The level of CLA-containing phosphatidylcholine (CLA-PC) increased dramatically in neurons incubated with CLA. Localization of BACE1 and APP in early endosomes increased in neurons treated with c9,t11 CLA concomitantly, the localization of both proteins was reduced in late endosomes, the predominant site of APP cleavage by BACE1. BACE1 and γ-secretase activities were not directly affected by c9,t11 CLA. CLA treatment did not affect the level of β-site APP-cleaving enzyme 1 (BACE1), a component of active γ-secretase complex presenilin 1 amino-terminal fragment, or Aβ protein precursor (APP) in cultured neurons. We found that c9,t11 CLA significantly suppressed the generation of Aβ in mouse neurons.
However, the effect of CLA on the generation of neurotoxic amyloid-β (Aβ) protein remains unclear. Two common conjugated linoleic acids (LAs), cis-9, trans-11 CLA (c9,t11 CLA) and trans-10, cis-12 CLA (t10,c12 CLA), exert various biological activities.
9 Department of Applied Biological Science, Faculty of Science and Technology, Tokyo University of Science, Noda, Japan.8 Department of Neurocognitive Science, Institute of Brain Science, Nagoya City University Graduate School of Medical Sciences, Nagoya, Japan.7 Laboratory for Proteolytic Neuroscience, RIKEN Center for Brain Science Institute, Wako, Japan.6 Advanced Prevention and Research Laboratory for Dementia, Graduate School of Pharmaceutical Sciences, Hokkaido University, Sapporo, Japan.5 Department of Neuropathology and Neurosciences, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Tokyo, Japan.4 Molecular and Cellular Biochemistry, Graduate School of Pharmaceutical Sciences, Tohoku University, Sendai, Japan.3 Health Chemistry, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Tokyo, Japan.2 Laboratory of Neuroscience, Graduate School of Pharmaceutical Sciences, Hokkaido University, Sapporo, Japan.1 Biomedical Research Institute, National Institute of Advanced Industrial Science and Technology (AIST), Tsukuba, Japan.A preparative scale purification combining crystallization and one step enzymatic process were made to afford purified cis-9,trans-11 (94.2%) with total recovery of 31%. The highest isomeric excess of cis-9,trans-11 isomer on trans-10,cis-12 CLA and good esterification degree was observed when the esterification condition were as follows: CLA/2-propanol molar ratio, 1:5 water, 200% wt of CLA CCL, 24μg of CCL/mg CLA temperature 30̊C time of reaction 6h and when mixture enriched in cis-9,trans-11 CLA was used as a substrate for esterification. The influence of alcohol substrate, CLA/alcohol molar ratio, lipase amount, reaction temperature, pH and time of the reaction on the total esterification and cis-9,trans-11 isomer purity in ester fraction were examined. The effect of various parameters on the esterification process of an almost equimolar mixture of cis-9,trans-11 (44.0%), trans-10,cis-12 (42.5%) and other CLA isomers (13.5%) was studied. Lipase from Candida cylindracea (CCL) was highly selective towards cis-9,trans-11 isomer and was chosen to catalyze esterification of a mixture of two CLA isomers. A method for purification of cis-9,trans-11 isomer of conjugated linoleic acid (CLA) is described.
